Download Brochure
X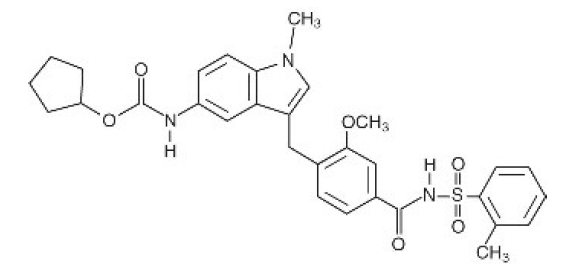
Zafirlukast (Amorphous) API
CAS Number: 107753-78-6
Zafirlukast (Amorphous) API
Therapeutic Category
Anti-Asthma
Respiratory
API Technology
Synthetic
Dose Form
Oral Solids
Dr Reddy's Development Status
Available
Available Regulatory Filing
USA
Mechanism of Action
Zafirlukast is a selective and competitive receptor antagonist of leukotriene D4 and E4 (LTD4 and LTE4), components of slow-reacting substance of anaphylaxis (SRSA). Cysteinyl leukotriene production and receptor occupation have been correlated with the pathophysiology of asthma, including airway edema, smooth muscle constriction, and altered cellular activity associated with the inflammatory process, which contribute to the signs and symptoms of asthma. Patients with asthma were found in one study to be 25100 times more sensitive to the bronchoconstricting activity of inhaled LTD4 than nonasthmatic subjects.
In vitro studies demonstrated that zafirlukast antagonized the contractile activity of three leukotrienes (LTC4, LTD4 and LTE4) in conducting airway smooth muscle from laboratory animals and humans. Zafirlukast prevented intradermal LTD4-induced increases in cutaneous vascular permeability and inhibited inhaled LTD4-induced influx of eosinophils into animal lungs. Inhalational challenge studies in sensitized sheep showed that zafirlukast suppressed the airway responses to antigen; this included both the early-and late-phase response and the nonspecific hyperresponsiveness.
In humans, zafirlukast inhibited bronchoconstriction caused by several kinds of inhalational challenges. Pretreatment with single oral doses of zafirlukast inhibited the bronchoconstriction caused by sulfur dioxide and cold air in patients with asthma. Pretreatment with single doses of zafirlukast attenuated the early-and late-phase reaction caused by inhalation of various antigens such as grass, cat dander, ragweed, and mixed antigens in patients with asthma. Zafirlukast also attenuated the increase in bronchial hyperresponsiveness to inhaled histamine that followed inhaled allergen challenge.
Indication
ACCOLATE is indicated for the prophylaxis and chronic treatment of asthma in adults and children 5 years of age and older.
Dr. Reddy's Expertise
Headquartered in Hyderabad, India, Dr. Reddy's Laboratories is one of the leading Active Pharmaceutical Ingredients (API) manufacturers and suppliers globally for Zafirlukast (Amorphous) API. Dr. Reddy's API business is a preferred partner to pharma companies across the US, Europe, Brazil, Latin America, Japan, China, Korea, Middle East and other emerging markets.
Dr. Reddy's API business thrives on the deep technical strengths established over the last 30+ years in the development and manufacture of complex APIs such as steroids, peptides, complex long chain molecules and highly potent APIs (HPAPIs / oncology drugs). This expertise is complemented by our prowess in intellectual property and regulatory affairs which helps us consistently meet and exceed regulatory standards. Dr. Reddy's Zafirlukast (Amorphous) API is the outcome of the extensive expertise in R&D, IP, and Regulatory.
A key component in helping our customers be first to market is a responsive supply chain. We achieve this by making sure that all our facilities are operating efficiently and to the latest standards of quality, safety, and productivity. A strong interconnect between business and factories allows for a quick reaction to dynamic market changes, so that we can avert shortages and meet sudden surges in demand.
Disclaimer
No information in this catalog - including any reference to any product or service - constitutes an offer for sale, or be construed as representing an offer for sale. Products protected under valid patents are not offered or supplied for commercial use. However, the research quantities of such products may be offered for the purpose of regulatory submissions, wherever such regulatory exemptions exist. The buyers should make their independent evaluation of the patent scenario for their respective markets and will be responsible for all patent related liabilities. Products protected under valid patents in India are not available for commercial use but would be available for Section 107A purposes.
Insights Delivered
Sign-up for our email service to get Market and Product insights and updates right to your digital doorstep
The categories of personal information collected in this form include name, company, and contact information etc. The personal information collected will be used for exploratory discussions on contract manufacturing, marketing and to perform research and analytics and others. For more information about the categories of personal information collected by Dr.Reddy's and the purposes for which Dr.Reddy's uses personal information, visit https://api.drreddys.com/privacy-policy.


